A Complete Modelling Approach for Battery Thermal Runaway Cell Heat Release, Venting and Particle Release
Published on February 04, 2026 · 2 min read
A complete thermal runaway workflow from the inception of heat release to the venting of gas and particles is developed in this work. The venting gas is usually formed due to the evaporation of the liquid electrolyte and thermal runaway reactions of the battery active material. As the battery active material undergoes physico-chemical transformation during the thermal runaway progression, proportionately the venting gas and particles are produced. Thermal runaway is kinetically evaluated in this work assuming a 0D lumped modelling. The evaporation of the liquid electrolyte is modeled using a Tanasawa phase change model, with saturation properties obtained from the PDB materials database. Each thermal runaway reaction may produce several gaseous components. For e.g., SEI decomposition produces C2H4, CO2 and O2. The fractions of these venting gas components produced from each thermal runaway reaction need to be prescribed by the user, are then correlated to the thermal runaway process for the production of the respective venting gases. An empirical correlation is formulated for the mass of generated particles. These input fractions usually are obtained from a testbed or literature analysis. Full modeling details are obtained from literature [1].
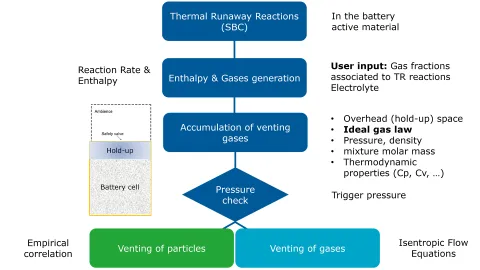
The modeling workflow depicted in Figure 1 is outlined below:
- Thermal runaway reactions in the battery active material
- Evaporation of the electrolyte
- Subsequent enthalpy and gases generation in the battery material
- Accumulation of the venting gases into a constant volume head-space
- Evaluation of the pressure, density, dynamic changes in the composition and the thermodynamical properties in the head-space. The gas composition in this head-space follows the ideal gas law.
- The pressure in the hold-up volume is compared against the prescribed trigger pressure for valve opening.
- The mass flow rate of the venting gas, calculated using the isentropic nozzle flow equations.
- Venting of gas and associated enthalpy transfer to the neighboring fluid domain
- Particle ejection into the fluid domain
To date, the modeling approach for thermal runaway has primarily relied on testbed data. This data serves as input for parameters such as heat generation rate, vented gases, and particle ejection. While this method is well-established and widely accepted, it is inherently dependent on the physical availability of the cell. Moreover, the required testing is both costly and destructive, which limits early-stage development and necessitates additional testing when a different cell is introduced. This approach remains appropriate when internal cell details are unknown and are expected to remain valid for a considerable period.
However, in recent years, more research is spent on understanding thermal runaway on a deeper level and providing modeling approaches that do not rely on testbed data as much. This is where the coupled approach comes into play.
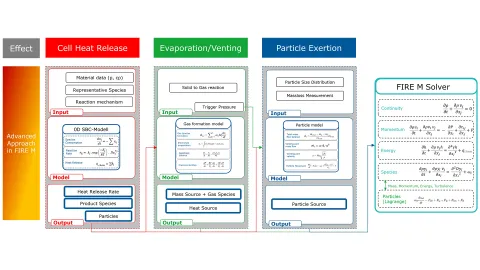
Figure 2 presents a flow chart illustrating the governing equations and sub-models: cell heat release in red, venting in green, and particle ejection in blue. The cell heat release is modeled through chemical reactions, and the resulting product species are used to determine venting gas fractions via dedicated sub-models. This coupling of heat release and venting forms a key cornerstone of the model, distinguishing it from previous measurement-based approaches. In addition, particle mass ejection is calculated. All resulting inputs are then integrated into our 3D CFD solver, ensuring that the interactions are fully captured.
This approach represents a significant step toward reducing reliance on testbed data. It introduces a software feature that will enable more accurate thermal runaway modeling and enhance the overall appeal of our software. However, AVL is more than just software. To demonstrate the robustness of this model, we must validate it through practical application—this will be detailed in the following section.
Test Case
The model is first evaluated using a simple representative battery module consisting of three prismatic cells, each measuring 173 × 125 × 45 mm, as shown in Figure 3 (a). The cells are placed inside an air-filled container and are referred to as Cell 1, Cell 2 (middle), and Cell 3. In the simulation, Cell 2 is uniformly heated until it reaches thermal runaway, which subsequently induces thermal runaway in Cell 1 and Cell 3 (see Figure 3 (b)). The thermal-runaway progression is modeled in 0D and is represented using the mechanism described by Ren et al. [2], consisting of six decomposition reactions of the battery active material components. This model is applied to all three cells.
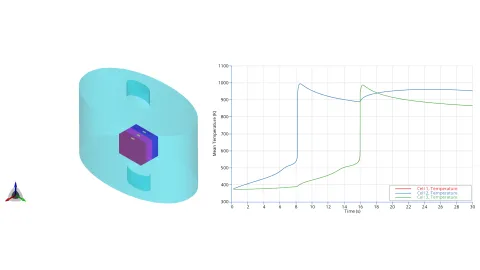

Figure 3: Test case geometry (top left) and mean temporal profiles for temperature (top right), battery active matter
The venting-gas ejection is illustrated in Figure 4. The animation displays the venting-gas velocity in Figure 4 (a) along with the methane species fraction in Figure 4 (b). As shown, the velocity reaches several hundred meters per second at the peak of the thermal-runaway event. As the current model employs a 0D solid body chemistry (representation of thermal runaway progression), the resulting gas velocities, mass flow rates and pressures tend to be over-predicted, and its applicability is therefore limited for e.g., to uniformly heated cells.
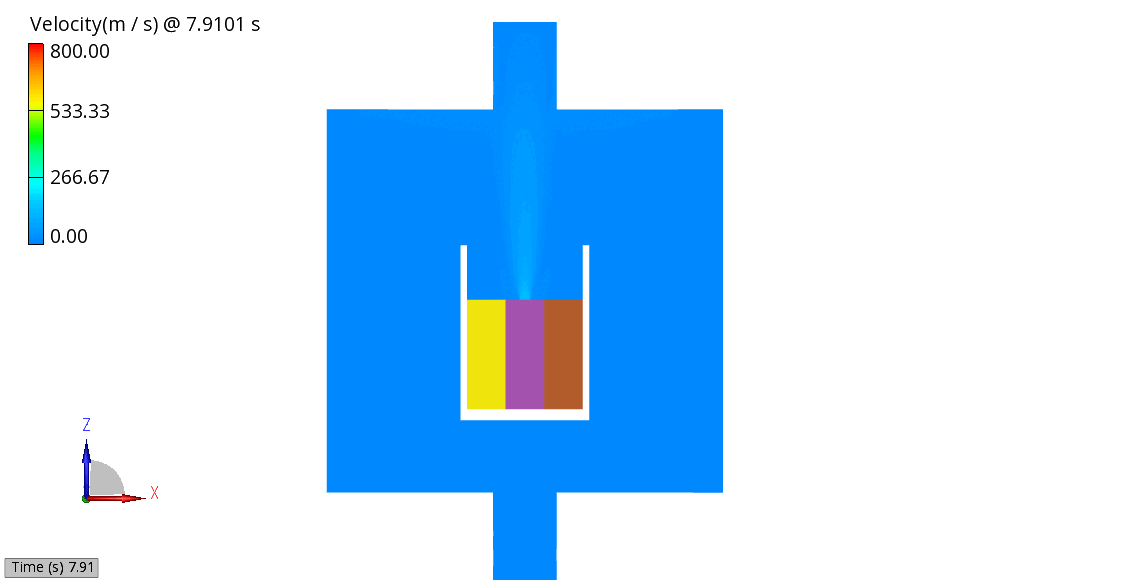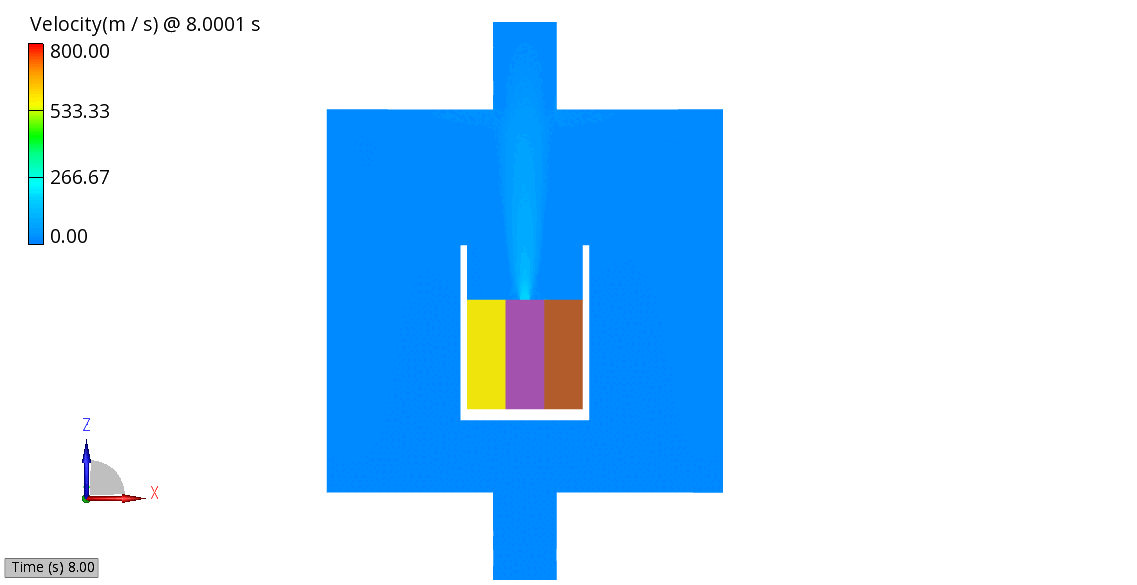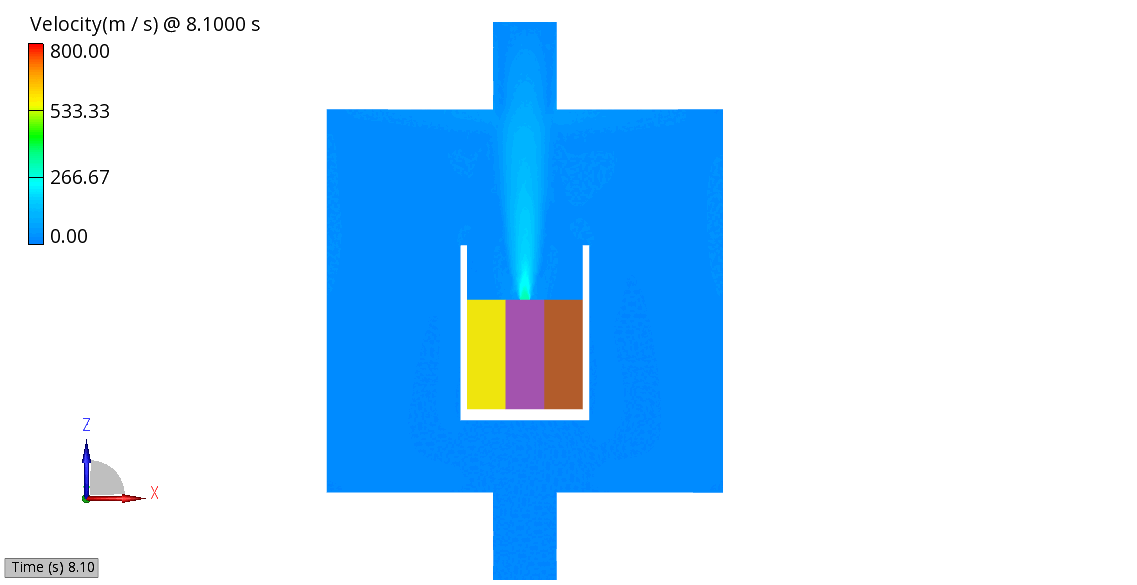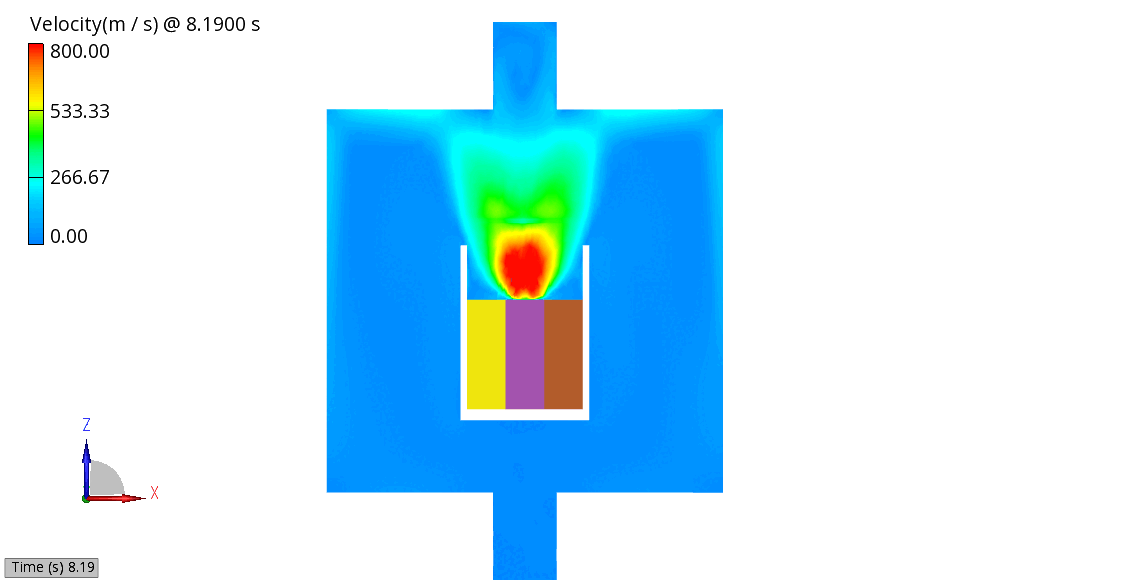
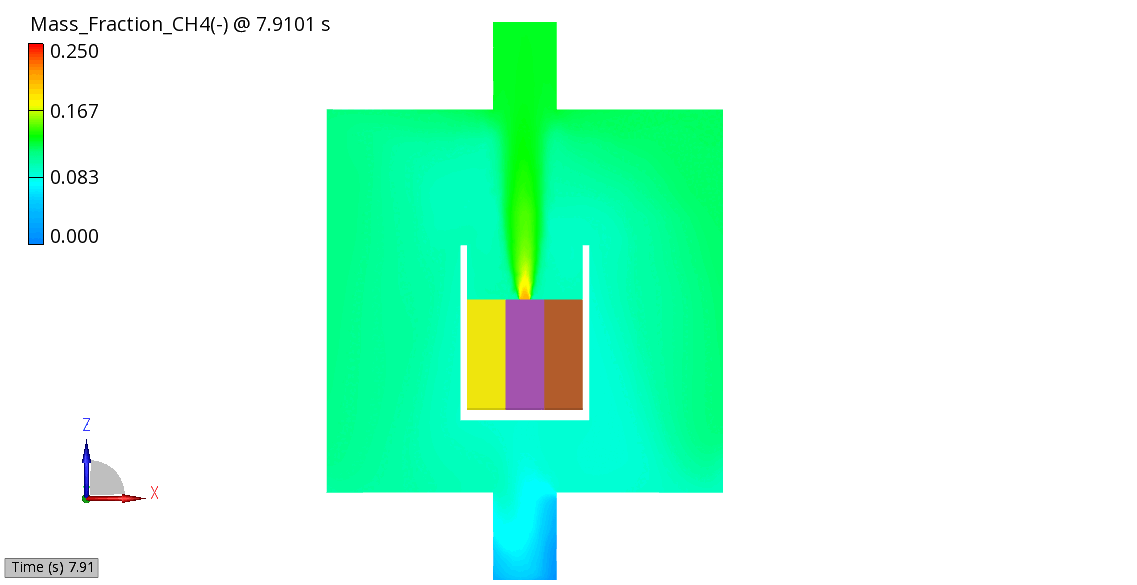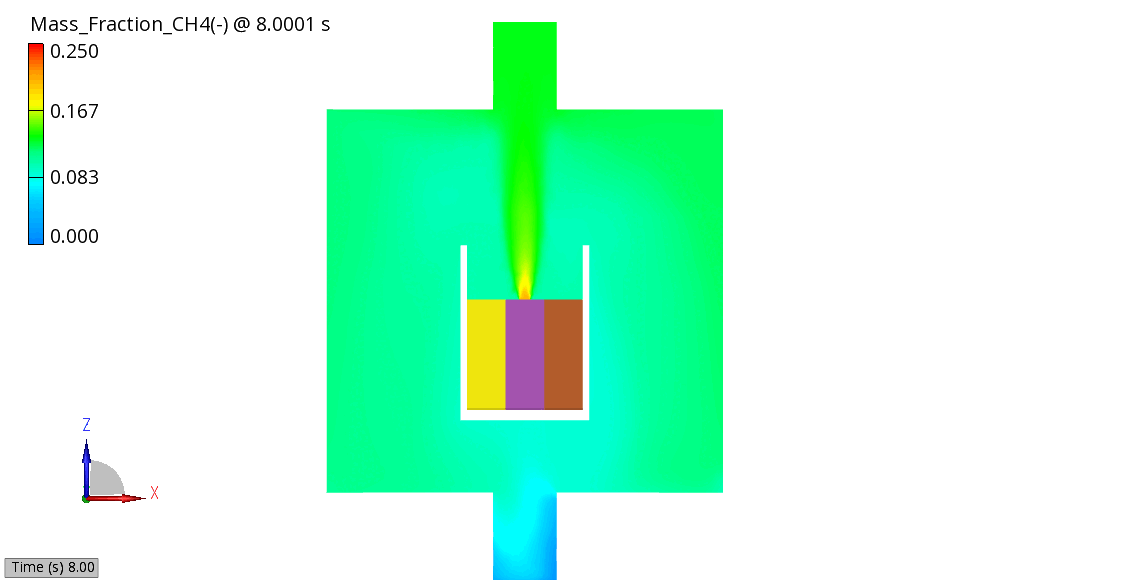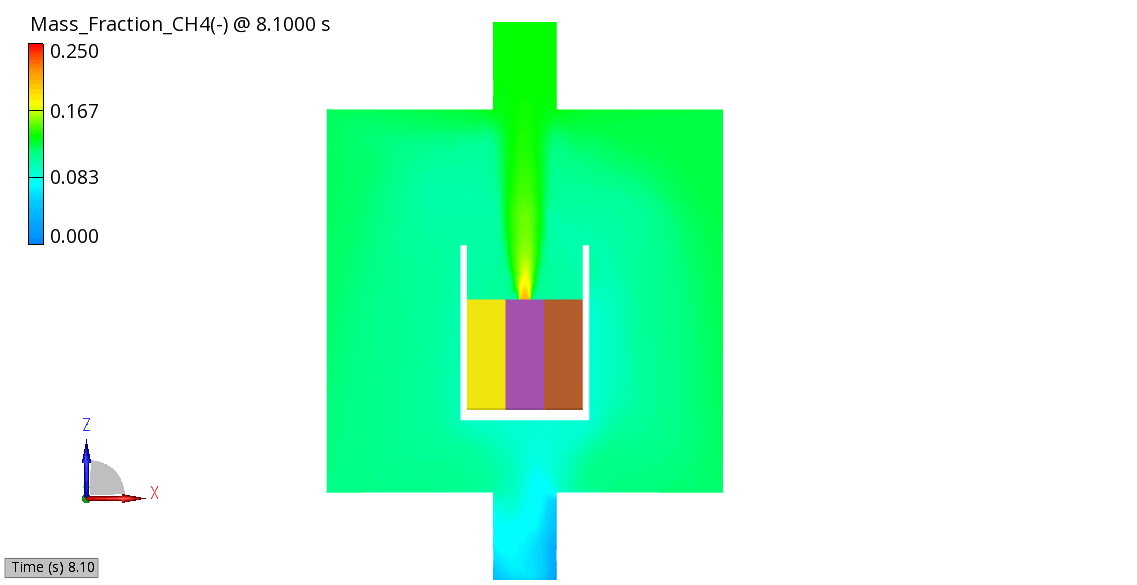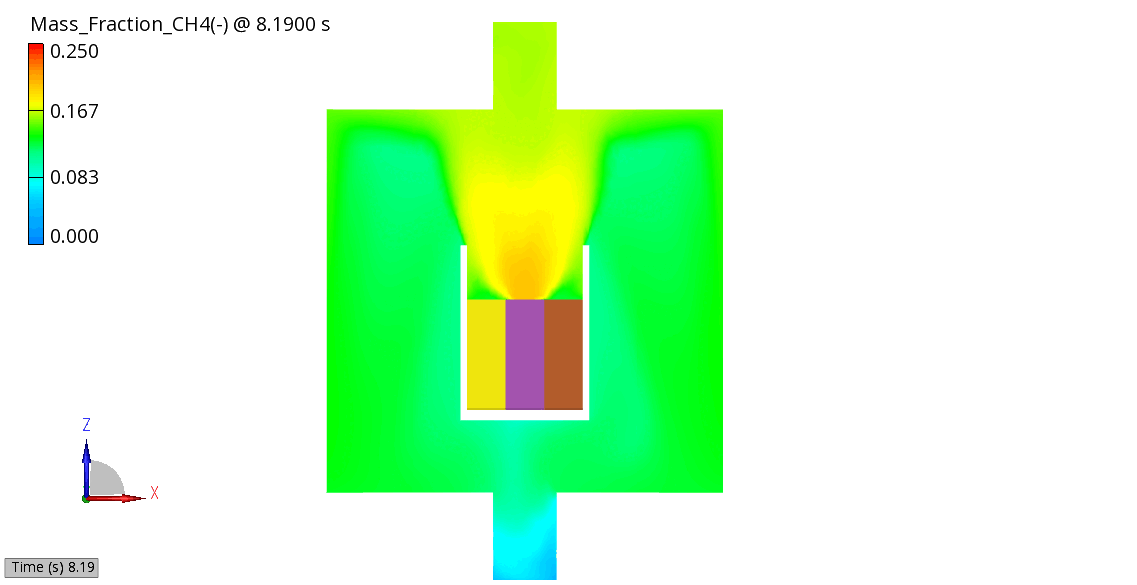
(b) CH4 Mass Fraction
Figure 4: Venting Gas Velocity and CH4 Concentration in the Venting Gas
An overview of how the workflow looks like for this model is shown in the Figure 5. However no model is capable of fully depicting reality – therefore it is important to us to also test the model and test it on a real battery thermal runaway event.
Currently we are putting our model to the test. Together with Virtual Vehicle we are working on a calibration workflow to set up the model for a selected NMC pouch cell. It is important for us to get a feel for the calibration needs, as well as to see the features benefits and limitations of this approach. Furthermore this gives us a default data set for one selected battery cell that we can then also use in our software to provide the user with proper starting values.

The current status of the calibration is well progressed. We are able to get much more insight with this model than before. Have a look at Figure 6 below. You can see a thermocouple instrumented on the battery cell as a black, dashed line. The red line is the FIRE M simulation and it is able to depict the temperature increase. What is however much more valuable is the 3D-cut through the whole simulation. We can see the cell (center) between two steel plates and it starts to eject the venting gas to the right. It does so during electrolyte evaporation when the temperature rise is not critical but also during full thermal runaway when the temperature rises several hundred degrees. Both of these effects can be captured with the model!

A fully coupled modeling approach that integrates cell heat release, gas venting, and particle ejection into a single workflow is introduced with the latest FIRE M release. This moves beyond traditional methods that rely heavily on testbed measurements, enabling predictive simulations based on minimal testbed data, reaction kinetics and material properties. The model is tested initially on representative battery modules and in some cases observed over-prediction of venting gas velocity and holdup pressure. Currently, the model is being tested with a selected NMC pouch cell to understand calibration needs and evaluate its benefits and limitations. The calibration process has progressed well, providing more insights than before, and the model can capture both electrolyte evaporation and full thermal runaway events.
Further Improvements
To address the over-predictions (e.g., flow rate and pressure) seen in test cases, further model development is required, since it currently relies on 0D solid-body chemistry for thermal runaway prediction.
In addition, extending the model’s applicability (e.g., to non-homogeneous systems) and improving 0D predictions may require incorporating 3D solid-body chemistry.
References
Wang et al, Revealing particle venting of lithium-ion batteries during thermal runaway: A multi-scale model toward multiphase process, eTransportation, Volume 16, 2023, 100237, ISSN 2590-1168, https://doi.org/10.1016/j.etran.2023.100237.
Ren et al, Model-based thermal runaway prediction of lithium-ion batteries from kinetics analysis of cell components, Applied Energy, Volume 228, 2018, Pages 633-644, ISSN 0306-2619, https://doi.org/10.1016/j.apenergy.2018.06.126.
Stay tuned
Don't miss the Simulation blog series. Sign up today and stay informed!
Read More About This Topic
Stay tuned for the Simulation Blog
Don't miss the Simulation blog series. Sign up today and stay informed!